
This video also goes through some notes on reactivity trends. This video will go through patterns of reactivity for group 1. A chart of the reactivity series of common metals is provided below. Students can also attempt to ionize an atom by removing its valence electrons. By choosing elements from the periodic table, atoms can be selected for a side by side comparison and analysis. Four major factors affect reactivity of metals: nuclear charge, atomic radius. In this simulation, students can investigate the periodic trends of atomic radius, ionization energy, and ionic radius. The elements within the same group of the periodic table tend to exhibit similar physical and chemical properties. It can also be used to obtain information on the reactivity of metals towards water and acids. 7.7: Group Trends for the Active Metals is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. Reactivity generally increases from the centre of the periodic table to the left (recall the right-hand side has non-metals), and also from the top of the periodic table down. The data provided by the reactivity series can be used to predict whether a metal can displace another in a single displacement reaction. 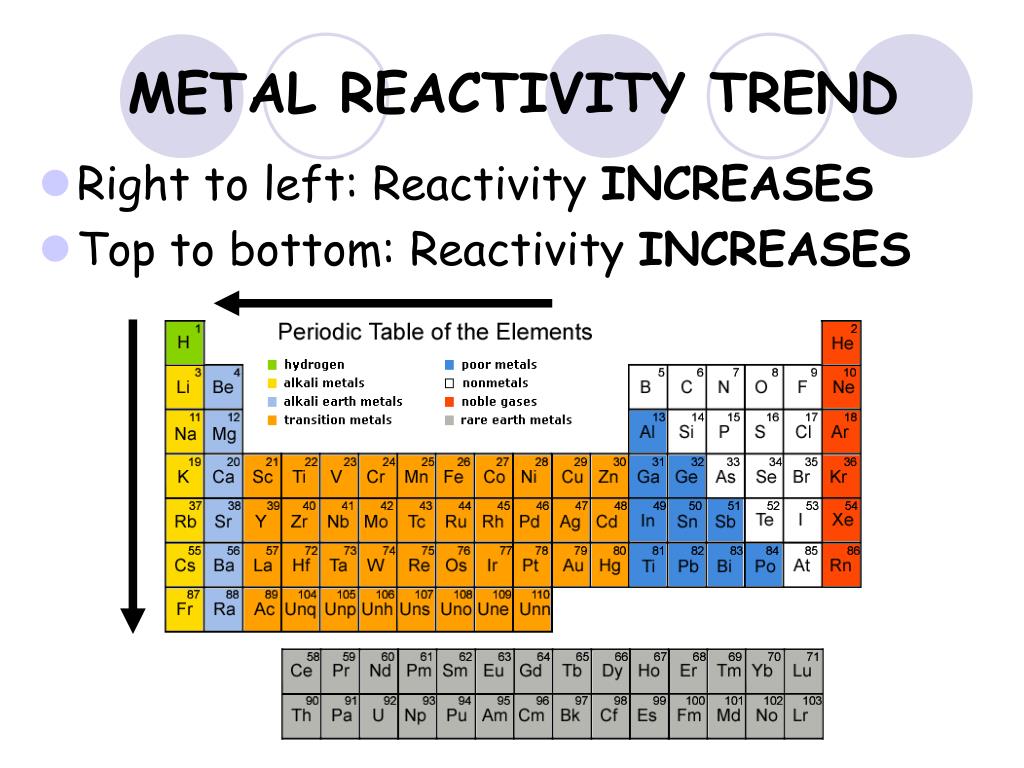
As a result the noble gases exist as single atoms and do not bond with other atoms. Trends in the Periodic Table Reactivity Stile Education 10.3K subscribers Subscribe Subscribed 2. See if you can apply your knowledge of the ionisation energy (energy required to expel an electron from an atom), electronegativity (ability to attract electrons to atom) and atomic radii (half the distance between two neighbouring nuclei of the same element) trends on the periodic table. The elements in group 0 (also referred to as group 8) of the Periodic Table are called the noble gases.They are chemically inert, meaning they do not undertake chemical reactions with other atoms. Metal reactivity relates to ability to lose. Four major factors affect reactivity of metals: nuclear charge, atomic radius, shielding effect and sublevel arrangement (of electrons). This video will go through some general reactivity trends across the periodic table. The elements within the same group of the periodic table tend to exhibit similar physical and chemical properties. Analyse patterns in metal activity on the periodic table and explain why they correlate with, for example:.This post will go through some patterns in metal activity for Prelim Chemistry. It can be used as a differentiated activity for the more able students within a group. This could be used to follow up some work on the periodic table where the trends in reactivity in groups 1 and 7 have been identified. What Are Some Patterns in Metal Activity? Chemistry for the gifted and talented: trends in reactivity in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
